 |
LigASite database of binding sites |
|
PDB ID and HEADER, TITLE and
COMPND records of the PDB file. | | (click anywhere in this window to remove it) |
|
| 1o2a |
|
|
| CRYSTAL STRUCTURE OF THYMIDYLATE SYNTHASE COMPLEMENTING PROTEIN (TM0449) FROM THERMOTOGA MARITIMA WITH FAD AT 1.8 A RESOLUTION |
TITLE |
|
|
| THYMIDYLATE SYNTHASE THYX |
COMPND |
|
|
|
|
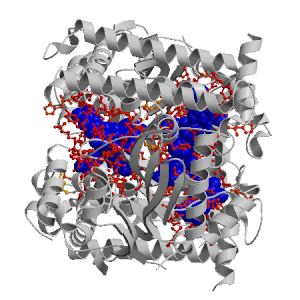
Figure showing the binding site residues. Ligands are displayed as
CPK. Figures were drawn with
Molscript (7) and rendered with
Raster3D (8). PISA coordinates
(3) are used when available
(all entries except NMR). Ligands do not appear on the picture when
PISA fails to apply symmetry operations to ligand coordinates. | | (click anywhere in this window to remove it) |
|
 |
|
List of binding site residues detected in this holo-structure. Column 1 gives the position, coloured on a yellow-to-red scale depending on the number of holo-structures where the residue is in contact with a ligand.
Column 2 gives the identifier of the chain to which the residue belongs.
Column 3 gives the 3-letter amino acid code, coloured according to physico-chemical type. The binding residues in this holo structure are listed for each ligand independently, following the ligand's unique ID. Note that since PISA files are used whenever available, chain identifiers may differ from those in original PDB files. | | (click anywhere in this window to remove it) |
|
| FAD F 710 |
|---|
| 27 | C |
ALA |
| 53 | C |
HIS |
| 55 | C |
THR |
| 58 | C |
GLU |
| 78 | E |
ARG |
| 79 | E |
HIS |
| 80 | E |
ARG |
| 81 | C |
ILE |
| 82 | G |
ALA |
| 83 | G |
SER |
| 85 | G |
ASN |
| 86 | G |
GLU |
| 87 | G |
LEU |
| 88 | G |
SER |
| | 90 | G |
ARG |
| 163 | C |
ASN |
| 165 | C |
ARG |
| 166 | C |
SER |
| 169 | E |
ASN |
| 173 | E |
LEU |
| 174 | E |
ARG |
| 178 | E |
HIS |
| 179 | E |
ALA |
| FAD B 715 |
|---|
| 27 | G |
ALA |
| 53 | G |
HIS |
| 54 | G |
GLU |
| 55 | G |
THR |
| 58 | G |
GLU |
| | 78 | A |
ARG |
| 79 | A |
HIS |
| 80 | A |
ARG |
| 81 | G |
ILE |
| 82 | C |
ALA |
| 83 | C |
SER |
| 85 | C |
ASN |
| 86 | C |
GLU |
| 87 | C |
LEU |
| 88 | C |
SER |
| 90 | C |
ARG |
| 91 | C |
TYR |
| 163 | G |
ASN |
| 165 | G |
ARG |
| 166 | G |
SER |
| | 169 | A |
ASN |
| 173 | A |
LEU |
| 174 | A |
ARG |
| 178 | A |
HIS |
| 179 | A |
ALA |
| FAD H 705 |
|---|
| 27 | A |
ALA |
| 29 | A |
VAL |
| 53 | A |
HIS |
| 54 | A |
GLU |
| 55 | A |
THR |
| 58 | A |
GLU |
| 78 | G |
ARG |
| 79 | G |
HIS |
| 80 | G |
ARG |
| | 81 | A |
ILE |
| 82 | E |
ALA |
| 83 | E |
SER |
| 84 | E |
TYR |
| 85 | E |
ASN |
| 86 | E |
GLU |
| 88 | E |
SER |
| 90 | E |
ARG |
| 91 | E |
TYR |
| 163 | A |
ASN |
| 165 | A |
ARG |
| 166 | A |
SER |
| 169 | G |
ASN |
| 173 | G |
LEU |
| 174 | G |
ARG |
| | 176 | G |
ASP |
| 178 | G |
HIS |
| 179 | G |
ALA |
| FAD D 700 |
|---|
| 53 | E |
HIS |
| 54 | E |
GLU |
| 55 | E |
THR |
| 58 | E |
GLU |
| 78 | C |
ARG |
| 79 | C |
HIS |
| 80 | C |
ARG |
| 81 | E |
ILE |
| 82 | A |
ALA |
| 83 | A |
SER |
| 85 | A |
ASN |
| | 86 | A |
GLU |
| 88 | A |
SER |
| 90 | A |
ARG |
| 91 | A |
TYR |
| 163 | E |
ASN |
| 165 | E |
ARG |
| 166 | E |
SER |
| 169 | C |
ASN |
| 173 | C |
LEU |
| 174 | C |
ARG |
| 178 | C |
HIS |
| 179 | C |
ALA |
|
|
|
| PDB |
The Protein Data Bank |
| CSA |
Catalytic Site Atlas |
| PDBSum |
Overview of the macromolecular structure |
| CATH |
Protein Structure Classification |
| Scop |
Structural Classification of Proteins |
| Pfam |
Protein Families and Domains |
| UniProt |
Universal Protein Resource |
LIGPLOT (only on holo-pages) is hosted at the EBI. The LigPlot Jmol links point directly to the Jmol visualisation interface provided on the PDBSum page. Note that due to different software used, the atomic contacts of LigPlot and LigASite do not necessarily correspond. | | (click anywhere in this window to remove it) |
|
Links to external databases: LigPlot (hosted at the EBI):
|
|
Several files are provided for download: | • The XML file defining the residue-ligand contacts; this file contains data on the apo and all holo-structures. |
| • The XML Schema file defining the semantics of the XML file |
| • 3D coordinates of the structure used in constructing LigASite (PISA structure file whenever available, PDB file otherwise. |
| • 3D coordinates of the combined binding residues in the apo structure |
| • 3D coordinates of the binding residues of the holo structure (only on the holo page) |
Coordinate files are in PDB format. | | (click anywhere in this window to remove it) |
|
|
|
|
|
List of related structure, containing both the apo-structure
and other holo-structures.
Column 1 gives the PDB ID and column 2 the unique ID
of the ligands (holo-structures only).
Clicking the blue 'Hide table of related structures' button
removes the entire table. | | (click anywhere in this window to remove it) |
|
|
| pdb ID |
|---|
| 1o24 |
|
Details |
|
| pdb ID |
Ligand Unique ID |
|---|
| 1kq4 |
FADB_315 FADH_305 |
Details |
|
FADD_300 FADF_310 |
| 1o2b |
FADE_600 FADH_610 |
Details |
|
FADC_615 FADK_605 |
| 1o26 |
PGEL_640 FADJ_610 |
Details |
|
FADB_615 PGEQ_635 UMPO_613 UMPH_618 PGEL_640 UMPK_608 FADN_605 FADG_600 PGEE_620 PGEP_625 UMPC_603 FADJ_610 |
|
UMPO_613 PGEP_625 FADJ_610 |
| 1o27 |
BRUF_618 FADK_605 FADB_615 |
Details |
|
BRUI_608 FADK_605 FADB_615 |
|
BRUC_603 FADE_600 FADH_610 BRUL_613 |
| 1o29 |
FADE_800 UFPC_803 UFPL_813 FADH_810 |
Details |
|
FADB_815 UFPI_808 UFPF_818 FADK_805 |
|
UFPL_813 FADH_810 |
| 1o28 |
EPED_700 UFPB_703 |
Details |
|
EPEC_710 UFPP_713 |
|
EPEK_715 PGEJ_735 UFPI_718 |
|
EPEN_705 UFPM_708 |
|
PGEG_725 |
| 1o25 |
UMPH_613 |
Details |
|
UMPF_608 |
|
UMPB_603 |
|
UMPD_618 |
|
|
|
Ligands present in this holo structure.
Column 1 shows the ligand HET code
Column 2 shows the name, chemical formula and (non-stereo) SMILES string.
Data in column 2 appears as 'not_found' when it is not present in the file
'pdb2smiles.xml' from www.rcsb.org. | | (click anywhere in this window to remove it) |
|
| FAD |
NAME: |
FLAVIN-ADENINE DINUCLEOTIDE |
|
FORMULA: |
C27 H33 N9 O15 P2 |
|
SMILES: |
Cc1cc2N=C3C(=O)NC(=O)N=C3N(CC(O)C(O)C(O)COP(O)(=O)OP(O)(=O)OCC4OC(C(O)C4O)[n]5cnc6c(N)ncnc56)c2cc1C |
|
|
|
|
v9.2
January 2011 |
Interdisciplinary Research Institute, Computational Biology, Villeneuve d'Ascq, France
University College London, Biomolecular Structure and Modelling Unit, London, UK
Hospital for Sick Children and University of Toronto, Structural Biology and Biochemistry Program, Toronto, Canada |
| Script execution time: 2.7904 seconds |

